INTERNATIONAL SYMPOSIUM Recycling and Reuse of Glass Cullet
University of Dundee 2003
ABSTRACT. The basic methods of recycling glass cullet are discussed in terms of the chemical engineering. Thus glass cullet is considered as a unified mineral raw material with a complex of inherent properties. The cullet of various fractional compositions should have various optimum reuse technologies. Considering glass cullet as a unified raw source allows us to create the reuse technology especially for this material, instead of recycling glass as an additive in some other technologies. Peculiar characteristics of the crystalline foamed glass technology using glass cullet as a raw material are discussed in the paper. After revealing physical and chemical properties of glass cullet as a separate raw material, its characteristic properties in disperse and monolithic conditions as well as in hydro- and pyroplastic states have been found. As a result, the method of recycling glass cullet taking into account its behaviour under various conditions has been offered and validated. It is possible to produce crystalline foamed glass in the form of blocks, gravel and decorative material. One of the most interesting features of the offered technology is absence of hydrogen sulfide in the products, while it is usually present in ordinary foamed glass. Besides, in the proposed technology there is no heat treatment of powder mixtures. Instead, the prepared powders aggregate into raw pieces before being heat-treated in a furnace. Making use of the discussed properties of glass cullet, the optimum ways of recycling it into commodities have been developed and realized. The products made from glass cullet as a raw material have the advanced construction and thermo-insulation consumer properties.
Keywords: Recycling, Glass cullet, Raw material, Heat-insulation, Construction, Technology.
INTRODUCTION
Glass cullet being one of the main components of solid domestic waste negatively affects the environment because a number of substances leach from it. Besides, glass cullet renders high traumatological effect on living organisms. Glass is the most stable component of wastes and is practically decomposable in natural conditions. Therefore, glass cullet has accumulated in an environment in quantities of tens millions tons and it is comparable to natural geological resources. From this point of view, glass cullet can be considered as a mineral resource – an amorphous silicate material of anthropogenic origin. The production of natural silicate materials implies significant consumption of material and power resources as well as unfavorable environmental effect of mining and processing mineral resources. Nevertheless, the anthropogenic source of silicate materials – glass cullet – is used in extremely limited quantities.
The up-to-date principles of utilization or recycling of glass cullet are mainly based on using it as a component of different manufacturing processes, for example, in fusion of glass, its fusion with cement clinker, its addition to concretes as a filling material, etc. Existing methods of glass recycling are incomplete, since for creation of an accomplished technology cullet should be considered as a raw material and its properties should be taken into account. After making a review of the methods of reuse of glass cullet [[1], [2]], all the methods have been classified into 4 groups according to the physicochemical properties of glass and its surface area[[3]], as shown in Table 1.
Table 1 The main ways of reuse of glass cullet depending on the glass properties
| Sizes of glass particles and properties of glass as a specific material | Areas of use | Component in the product replaced by glass |
| Lump glass, fragments larger than 0,5-1 mm. High strength, good adhesion to silicate materials, high decorative properties | Underlying layer for roads (underlayers, drainages, backfills) Decorative elements Lumpy filling agent in concrete Glass - asphalt |
Crushed stone, gravel |
| Coarse-dispersion powder, fragments from 0,05-0,1 up to 0,5-1,0 mm. High abrasive-resistance, high chemical stability | Abrasive element Friction material for matches and ammunition Filtration medium Component for forms in a foundry practice |
Quartz sand, emery, grit of metals, oxide and metal fraction, scoria |
| Middle and highly dispersion powder dust, fragments less than 0,1 mm. Ion-active properties of surface, astringent properties | Filler in a paints and plastics Absorbent and cation-exchange material Addition component in a cement clinker before and after calcination Additional component for cement |
Natural clay, zeolites, carbonates, fly ash, hydrate of calcium silicate, replacement of Portland-cement or puzzolan |
| Pyroplastic properties of glass at temperatures higher than 680-850 °C | Binder in ceramics and brick Decorative and facing materials Heat-insulating materials of filamentary or foam structure |
Mineral connecting (such as clay), scorias and fusible silicates |
Such classification is based on the differences in the properties of glass having different dispersion. It is especially important that with the increase of the glass dispersion the methods of its processing based on the surface characteristics of the material begin to predominate. And the most relevant property of a glass surface is its cation-exchange activity, implying washing away of sodium ions and the formation of polysilicon acids on the surface. At high temperatures, glass as a thermodynamically unstable supercooled liquid possesses two technologically essential characteristics. First, it has a wide temperature softening range. Second, it tends to crystallise. These properties of glass as an individual material and a mineral source make it possible to develop the technology for glass cullet as the main raw material.
It is important that industry, which could consume the mentioned amounts of glass cullet, should have a considerable need in a silicate raw material. Evidently, the industry of construction materials satisfies this condition. It is also obvious from Table 1 that practically all the offered methods of glass reuse are somehow connected with the production of building materials. Analysis of the construction materials market shows that in this area there is the greatest demand for heat-insulating materials, and a stable growth of this sector of the construction materials industry is expected.
Foamed glass is the most promising heat-insulating material even in spite of rather a high price of a volumetric unit. Therefore, the reuse of glass cullet for the production of foamed glass would be an optimum solution of the problem, both from the ecological and economical points of view. In this connection, we will consider some peculiarities of producing this material.
RESULTS AND DISCUSSION
Powdered Sulphate Technology
Foamed glass (cellular glass) is a cellular heat-insulating material, produced by sintering of glass powder dust at high temperatures with simultaneous foaming of it under the influence of gassing agent. The technological process of foamed glass production, according to the traditional powder technique, has the following main operations. The first stage is glass manufacture from quartz sand, chalkstone and soda salt. The obtained glass is crumbled up, mixed with a gas developing agent (1-5 %) and loaded into forms made of a heatproof material. Forms are heated up, the glass sinters and is foamed by gaseous products of the reaction. After cooling, the material with a cellular structure results. producing of initial glass is absent, when glass cullet or glassy rocks are used as a raw material.
The chemical essence of gassing proposed by F.Shill [[4]], may be expressed as follows:
glass-SO42-+2C -> glass-S2-+ CO + CO2
The obtained material contains a fair quantity of hydrogen sulphide in its cells and, moreover, it is capable of evolving this gas when the sulphides contained in glass get in contact with the moisture of the air. It limits the fields of its application and makes the technology unattractive from the ecological point of view. However, it is possible to synthesise foamed glass without sulphate sulphur in its initial composition. Demidivich [[5]] came to the conclusion, that with the heat treatment of the initial composition, the evolving of gases can be provided by the oxidation of carbon by steam:
C + H2O -> CO + H2 and C + 2H2O -> CO + 2H2 .
Therefore, for the oxidation of carbon in the system and foaming of the material it is necessary to provide the evolving of steam at temperatures 600-850 °C. Only chemically fixed water can be such a source. These hydroxyl groups can enter the system at the stage of preparation of powders. Activity of the process of hydration of glass is proportional to the temperature and the surface area. Alongside with the hydration, the process of dissolution of glassy alkalinous silicates in water takes place, and it is accompanied by hydrolysis.
Thus, to produce foamed glass by the sulphur-absent technology, steam as an oxidant might be used, which may enter the system at the stage of preparation of raw materials, when with the hydrolysis of the dispersed glass powder hydroxyl groups appear on its surface. So we will consider the possibility of realisation of these principles in practice.
Steam-oxidation Foaming of Glass
For steam evolution in the system at foaming temperatures, it is necessary to introduce hydroxyl groups at the stage of preparation of the powder. The main role in the hydration of the dispersed glass is played by the exchange of the surface sodium ions of the material for the H+ ions of the water environment. Typical curves of titration of powders by an acid are represented in Figure 1.
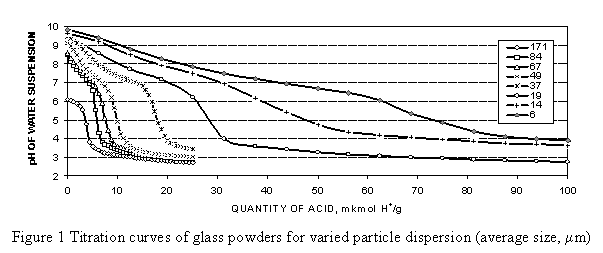
The process of titration of a various dispersion powder suspensions in water by an acid and alkali shows that these suspensions behave as a mild basic compound. A higher dispersion of the powder leads to the more basic properties of the suspension. It was found that the amount of sodium ions capable of the ion exchange with H+ ions is in inverse proportion to the square of an average radius of glass particles.
An amorphous layer depleted with Na+ ions covers the powder surface after the hydrolysis. It was shown [[6]] that with extraction at higher temperature (105-140 °C), calcium and sodium monosilicates result as a solid residue. In our opinion, at room temperatures the process of glass surface extraction is weaker and it limits the stage of ion exchange and forming of acids on the surface. A layer of polysilicic acids appears on the glass surface as a result of ion exchange and hydrolysis. These acids give astringent properties to the system due to the dehydration and coagulation of these amorphous acids. The displacement of pH value also proves that ion-exchange groups H+ of the surface have a different value of instability constants that is typical of the compounds with a variable composition, such as polysilicic acids.
It is confirmed by the data of scanning electron microscopy introduced on Figure 2. One can see acute edges on the fresh surfaces of glass particles and the absence of the amorphous layer which covers the surface of the glass particles subjected to hydrolysis and exposed at room temperature until strong composition is formed. Such an astringent property is well known and thoroughly studied [[7]], but it is possible to use it for formation of preforms for future foaming. Therefore, the hydrolysis of sodium polysilicons on the glass surface allows us not only to obtain the system prone to aggregation, but also to manufacture strong materials on the basis of glass powder. It is interesting from the practical point of view.

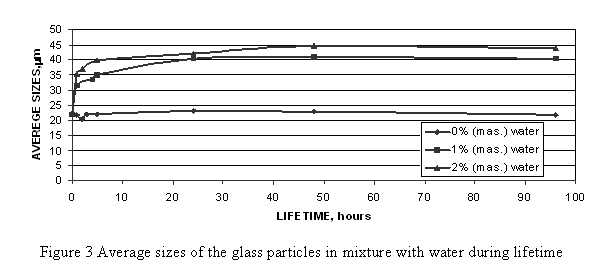
Glass powders in the presence of water undergo spontaneous aggregation in the initial period of interaction that is confirmed by data of sedimentation dispersion particles analysis (Figure 3).
Presence of water even in little quantities obviously results in fast aggregation of particles. This process leads to the growth of strength of compositions made of concentrated aqueous glass slurries. So the research of strength of the materials prepared by mixing of glass with water shows that growth of the material strength does not practically depend on the ambient humidity and takes place most intensively on the first day. It could be explained by the development of the process of formation and hardening of a gel-type layer on the glass surface. From the technological point of view, preparation of strong compositions from glass powder is important also because it allows us not only to obtain preforms handy for transportation and processing, but also to avoid glass dust which is typical of powdered processes.
Formation of polysilicon acids in the system results in evolution of steam at the temperatures above 500-600 °C at which glass is close to pyroplastic state. It makes it possible to use steam for the reactions of carbon oxidation.
The processes of gas generation can be described by the following scheme:
C + H2O = CO + H2; (1)
C + 2H2O = CO2 + 2H2; (2)
CO2 + C = 2CO; (3)
CO + H2O = CO2 + H2 (4)
Analysis of the dependence of the Gibbs function of the mentioned reactions on temperature shows that reactions (1) - (3) at the temperatures higher than 700 °C are spontaneous. Only reaction (4) is inhibited. In spite of the fact that the values of the Gibbs function for gas generation reactions 1-4 have a lower order, than the reactions of the sulphate oxidation, the process of foaming proceeds. The process doesn’t require a great amount of gases, besides, it can be controlled by regulating an amount of added coal which shifts the reaction equilibrium.
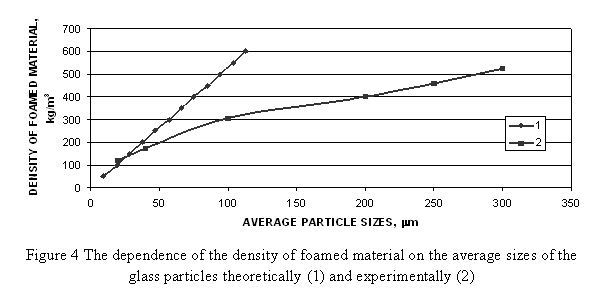
For estimation of the steam amount evaluated at indicated temperatures it is possible to use the published data on the temperature stability of hydroxyl groups on the glass surface [[8]]. It was shown, that at temperatures of glass softening about 700-800 °C, the concentration of Si—OH groups changes from primal 5 up to 1 group per 1 nm2, i.e. five times. Thus, it is suggested, that upon foaming the material one of five H2O molecules would take part in the gas generation reaction. Hence, using the experimentally obtained surface area of glass powder, it can be calculated, that for obtaining foamed glass with the density of 100 kg/m3 it is necessary to use hydrated powder of glass with an average particle size of 19 mkm as raw material, and for obtaining 200 kg/m3 - 38 mkm, accordingly. It is interesting, that the calculated dependence of the density of the material on the particle size of a glass shows good correspondence with the experimental data (Figure 4).
Experimental data shows that at each temperature of heating there is a particular period in which it is possible to reach the minimum of possible density, and the density varies a little with time. At low temperatures, low density of a material cannot obtained. At the temperatures more than 850 °C the further increase of temperature has no effect on density of the foamed glass. The presence of induction effect shows the evidence of the period that is necessary for warming of all the material and the heterogeneous reactions of gas generation begin.
From the chemical point of view the manufactured foamed material is similar to silicates such as glass but the physical nature of the material is closer to crystals that can be demonstrated by an X-ray structure analysis and its mechanical characteristics. Variation of conditions of obtaining allows us to vary the density of the material and its heat insulation properties (Table 2).
Table 2 Properties of produced foamed silicate material with standard densities
| Density, kg/m3 | Compressive strength, MPa | Heat permeability, W/(m·K) |
| 150 | 0,7 | 0,040 |
| 200 | 1,0 | 0,045 |
| 300 | 1,5 | 0,060 |
| 400 | 3,5 | 0,075 |
| 500 | 5,0 | 0,080 |
| 600 | 7,5 | 0,100 |
Usage of steam as an oxidant and the above mentioned oxidation-reduction reactions allow us to avoid precipitant gassing in the system. The material is characterised by the uniform structure of foam and thin baffle plates between cells. Regulating the conditions of raw material processing and heating, it is possible to obtain foam with the sizes of cells from 0,1-0,2 mm up to 5-7 mm (Figure 5).
Special Sorts of Foamed Glass
It is possible to produce foamed glass of different colours by adding different ions to the initial composition. Such foamed glass is not only a valuable heat-insulating material, but can also be used as a wonderful decorative material.

Besides, the offered technology implies using practically any kind of glass as a raw material. So, the experiments with glass cullet of a different origin have shown practically the complete identity of the products. Moreover, even such sort of a raw material as cathode ray tubes can be reused by this method, the mechanical separation of the details of tubes being not necessary. An interesting feature of the prepared foamed glass is not only its good thermal insulating property, but also its ability to absorb hard radiation, due to the presence of lead in the structure of glass. So, experiments have shown, that the attenuation constant of a X-radiation with the wavelength of 1,54A is 4,420 cm-1 for the material with the density of 210 kg/m3 and 0,045 W/(m·K) heat conductivity. The emission of lead from this material corresponds to the ecological norms. The characteristics of the obtained material enable it to be suggested for thermal insulation of atomic power stations and the objects with possible radiation.
The other important feature of the offered technology is the possibility of creation of preforms of different sizes according to the task. For the production of foamed glass blocks it is convenient to make large rectangular bars equal to the internal sizes of forms for heat treatment. And for the manufacture of granulated foamed glass it is possible to produce spherical preforms by the method of spherical pellets granulation, which are convenient for storage, transportation and foaming in a revolving furnace (Figure 6).
Experimentally it was found out, that the process of granulation is influenced by the humidity of the mixture and the dispersion of a glass powder. The maximum size of spherical pellets increases with the humidity of the mixture. However, the increase of humidity is expedient only in a particular range. Exceeding of this limit results in the destruction of granules.
The maximum size of spherical pellets grows in accordance with increase of a share of large particles in initial mixture. It was found that optimum moisture of the powder exists for each dispersion of a glass powder. At an optimal humidity of the mixture the formation of pellets is fast and with the least sizes dispersion of produced granules. The optimal humidity decreases by 1,45 % (mass) when the surface of the glass powder increases by 104 m2/kg. The rate of heating in the furnace noticeably influences the properties of granulated foamed glass. This parameter depends on the kinetics of heat- and mass-transference in spherical preforms and it is preferential to provide the maximum heating rates in the furnace. Therefore, this furnace is heated by a forward flow in relation to the flow of the material.

CONCLUSIONS
Glass cullet as a unified raw source allows us to create the reuse technology especially for this material. Thus, the obtained data shows, that the unsorted glass cullet can be utilized as a raw material for the production of heat-insulating materials. The produced materials having the cellular foamed structure possess high performance characteristics as construction materials that can be used as heat insulation in industrial and civil construction, pipe lines and process equipment.
REFERENCES:
- Meyer, C. Recycled glass – from waste material to valuable resource. The International Symposium on Recycling and Reuse of Glass Cullet, United Kingdom, Dundee, 2001, pp. 1-10.
- Pascoe, R. D., Barley, R. W. and Child, P.R. Autogenous grinding of glass cullet in stirred mill. The International Symposium on Recycling and Reuse of Glass Cullet, United Kingdom, Dundee, 2001, pp. 15-27.
- Kitaygorodsky, I. I. Technology of glass. Building Publisher, Moscow, 1967, 564 p.
- Shill, F. Foamed glass. Building Publisher, Moscow, 1965, 307 p.
- Demidovitch, B.K. Foamed glass. Science and Technology Publisher, Minsk, 1975, 248 p.
- Mavilia, L., Bella, G.Di. and Corigliano F. Characterization and valorization of the solid residue from glass extraction with an alkali solution. The International Symposium on Recycling and Reuse of Glass Cullet, United Kingdom, Dundee, 2001, pp. 55-61.
- Dyer, T.D. and Dhir R.K. Use of glass cullet as a cement component in concrete. The International Symposium on Recycling and Reuse of Glass Cullet, United Kingdom, Dundee, 2001, pp. 157-166.
- Curthoys, G., Davydov, V.Y. and Kiselev A.V. J. Collloid Interface Sci., 1974, 48, pp. 58-62.














